How GxP Compliance Solutions Improve Regulatory Control

In highly regulated industries like pharmaceuticals, biotechnology, and medical device manufacturing, maintaining compliance is not optional—it is a legal and operational necessity. This is where GxP compliance solutions play a critical role. From ensuring product quality to maintaining data integrity, these systems help organizations meet strict regulatory standards while improving efficiency.
In this guide, we will explore what GxP compliance means, how GxP software installation works, and why tools like CMMS calibration software are becoming essential in modern compliance environments.
Understanding GxP Compliance in Regulated Industries
GxP is an umbrella term that refers to “Good Practice” quality guidelines and regulations. The “x” stands for different fields such as Manufacturing (GMP), Laboratory (GLP), or Clinical (GCP).
At its core, GxP ensures that products are consistently produced and controlled according to quality standards. It focuses on:
- Product safety and efficacy
- Data integrity and traceability
- Proper documentation and validation
- Risk management in processes
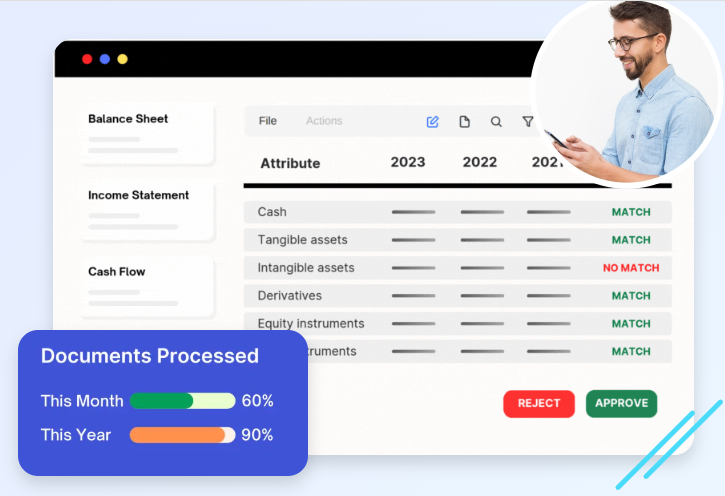
GxP compliance solutions are software and system frameworks designed to automate and manage these requirements. Instead of relying on manual documentation and fragmented processes, organizations use digital tools to maintain accuracy and consistency.
Why GxP Compliance Solutions Are Essential Today
The complexity of regulatory requirements is increasing globally. Authorities like the FDA, EMA, and WHO require strict adherence to compliance protocols.
Here’s why modern businesses rely on digital compliance solutions:
1. Reduced Human Error
Manual documentation increases the risk of mistakes. Automated systems reduce errors in reporting, validation, and recordkeeping.
2. Real-Time Data Tracking
GxP systems allow organizations to monitor operations in real time, ensuring immediate detection of deviations.
3. Audit Readiness
Inspection readiness is one of the biggest advantages. With structured digital records, audits become faster and less stressful.
4. Improved Productivity
Teams spend less time on paperwork and more time on value-driven tasks like research and production.
In short, GxP compliance solutions ensure that quality and efficiency go hand in hand.
GxP Software Installation: Step-by-Step Overview
Implementing compliance software is not just about installing a tool—it’s about integrating it into a regulated ecosystem. A proper GxP software installation process ensures validation, security, and regulatory alignment.
1. Requirement Analysis
The first step involves understanding organizational needs. This includes identifying compliance gaps, workflow requirements, and system expectations.
2. System Validation Planning
Validation is a key regulatory requirement. A detailed plan is created to ensure the software meets GxP standards, including Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ).
3. Installation and Configuration
The software is installed in a controlled environment. Configuration is done based on business workflows, user roles, and compliance rules.
4. Data Migration and Integration
Existing data from legacy systems is carefully migrated. Integration with ERP, LIMS, or CMMS systems ensures smooth data flow across departments.
5. Testing and Validation
Rigorous testing ensures that the system works as intended and complies with regulatory expectations. Any deviations are documented and resolved.
6. User Training and Deployment
Employees are trained to use the system effectively. Once approved, the system is deployed across the organization.
A well-executed GxP software installation ensures long-term compliance and reduces operational risks significantly.
Role of CMMS Calibration Software in GxP Compliance
Calibration and maintenance are critical in regulated environments, especially in manufacturing and laboratory settings. This is where CMMS calibration software becomes extremely important.
CMMS (Computerized Maintenance Management System) helps organizations manage equipment maintenance, calibration schedules, and asset tracking.
Key Benefits Include:
- Automated Calibration Scheduling: Ensures equipment is calibrated on time without manual tracking
- Regulatory Compliance: Maintains audit-ready calibration records
- Asset Lifecycle Management: Tracks equipment from installation to retirement
- Reduced Downtime: Prevents unexpected equipment failures through preventive maintenance
In GxP-regulated environments, even a small calibration error can lead to major compliance issues. CMMS tools ensure that all equipment remains within validated operational limits.
Key Features of Effective GxP Compliance Solutions
Not all compliance tools are created equal. High-quality systems share some essential features:
1. Electronic Documentation Control
Paper-based systems are replaced with secure digital document management, ensuring traceability and version control.
2. Audit Trails
Every action within the system is recorded, ensuring transparency and accountability.
3. Access Control and Security
Role-based access ensures only authorized personnel can modify critical data.
4. Validation Support
Built-in validation tools help organizations meet regulatory requirements efficiently.
5. Integration Capabilities
Modern systems integrate seamlessly with laboratory, manufacturing, and maintenance platforms.
These features ensure that GxP compliance solutions are not just tools, but complete ecosystems for regulatory management.
Challenges in Implementing GxP Systems
While the benefits are significant, implementation is not without challenges:
- High initial setup and validation costs
- Resistance to change from employees
- Complex integration with legacy systems
- Continuous updates to meet evolving regulations
However, these challenges can be managed with proper planning, training, and expert consultation.
Future of GxP Compliance and Digital Transformation
The future of compliance is increasingly digital. Technologies like AI, machine learning, and cloud computing are transforming how organizations handle GxP requirements.
We can expect:
- Predictive compliance monitoring
- AI-powered audit preparation
- Cloud-based validation systems
- Real-time global regulatory tracking
As industries evolve, GxP compliance solutions will become more intelligent, automated, and interconnected.
Conclusion
GxP compliance is no longer just about meeting regulatory requirements—it’s about building a culture of quality, safety, and efficiency. With the right GxP compliance solutions, organizations can streamline operations, ensure data integrity, and maintain full regulatory readiness.
Proper GxP software installation ensures systems are validated and compliant from day one, while tools like CMMS calibration software help maintain equipment reliability and operational accuracy.
In a world where compliance is closely tied to business success, investing in robust digital solutions is not just an option—it is a necessity.


 English
English